General Discussion
Related: Editorials & Other Articles, Issue Forums, Alliance Forums, Region ForumsFood Science Babe: 5 basic scientific concepts to help you spot pseudoscience
Here are 3. Go to link for the rest:
There is so much pseudoscience being spread around nowadays, and sometimes it can be hard to spot, especially for those who don’t have a background in science. A lot of it is based on a small amount of truth and by people who you may think you can trust, so it can be difficult to differentiate from good, truthful science.
These five basic scientific concepts can help you to spot pseudoscience when you see it:
#1) Everything is made of chemicals
You are made up of chemicals, and everything you eat is made up of chemicals. This series of posters by James Kennedy illustrates how all natural fruits are made up entirely of chemicals, some that may even have names you can’t pronounce and don’t recognize:

A “chemical free lifestyle” is impossible. Water and oxygen are examples of chemicals that you need to survive, and you wouldn’t last very long without them. So, the next time you see a food product that purports to contain “no chemicals” or a blog with products that you can purchase to live a “chemical-free lifestyle,” you can be sure that it’s purely pseudoscience because EVERYTHING IS MADE UP OF CHEMICALS.
#2) The dose makes the poison
“Toxic” is a word thrown around a lot on food blogs and in food marketing, but it’s almost always used incorrectly. There is no such thing as a “toxic chemical,” there are only toxic doses. The phrase “the dose makes the poison” applies to all chemicals, both natural and synthetic.
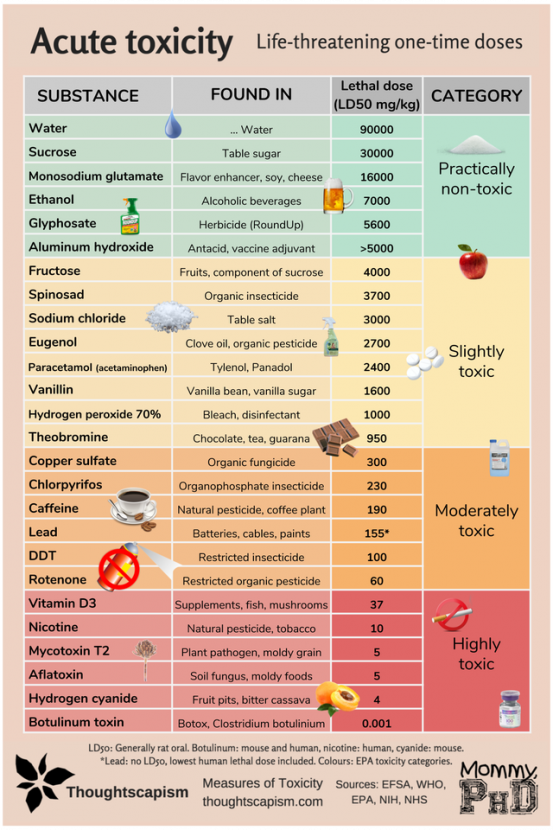
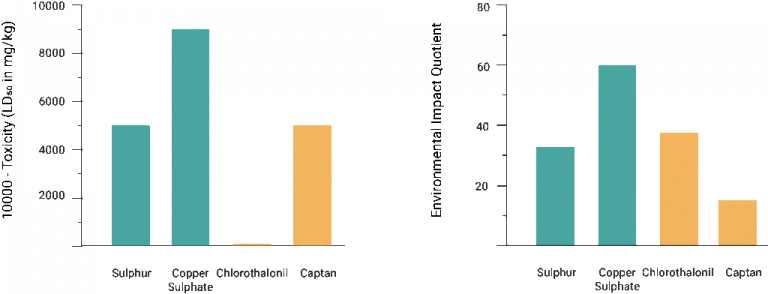
Acute toxicity measures lethal effects from a large one-time dose and is expressed as the LD50. The LD50, or median lethal dose, is determined experimentally, usually with rats or mice. It is the single acute dose that will kill 50 percent of a population given that dose. Chronic toxicity measures the lowest level of daily exposure that causes harm. Any chemical can be toxic, even water and salt, so products that state things like “no toxic chemicals” or “no toxic pesticides” don’t make any sense as every chemical can be toxic at a large enough dose. And, if someone tries to tell you that a food ingredient or a chemical is “toxic” without being able to tell you the concentration at which the chemical occurs in the food and the dose that is toxic, they either have no idea what they’re talking about, are trying to deceive you, and/or are trying to sell you something.
#3) Whether a chemical is natural or synthetic tells you precisely nothing about its safety
As stated previously, “the dose makes the poison” applies to both natural and synthetic chemicals, so whether a chemical is natural or man-made tells you precisely nothing about how dangerous it is. For example, sodium thiopenta, a man-made chemical, is used in lethal injections, but it’s about as toxic as naturally occurring amygdalin, which is contained in almonds and apple seeds. Similarly, many natural and organic pesticides have a lower toxic dose than their synthetic counterparts.
The appeal to nature fallacy is the belief that is what is natural must be good, and that which is unnatural must be bad. “Natural” has no definition per the FDA in regards to food labeling, yet it’s used in food marketing all of the time. You can be sure to ignore anyone or any product that uses the word “natural” to mean better or safer as the “appeal to nature fallacy’ is just that, a fallacy. It’s pure pseudoscience.

More: https://www.agdaily.com/insights/food-science-babe-5-basic-scientific-concepts-help-spot-pseudoscience/
ismnotwasm
(41,980 posts)stopdiggin
(11,306 posts)Hermit-The-Prog
(33,346 posts)From the article:
Here is a comparison between two organic fungicides (shown in teal) and two synthetic fungicides (shown in gold):
Sorry, but neither sulphur nor copper sulphate are organic; they are inorganic.
The point about the "appeal to nature fallacy" is still completely valid. Poison ivy, rattlesnake venom, and cat vomit are all natural, but they make a really nasty salad.
Quixote1818
(28,936 posts)For Sulfur it looks like there are both organic and inorganic types:
Protein and organic cofactors
Sulfur is an essential component of all living cells. It is either the seventh or eighth most abundant element in the human body by weight, about equal in abundance to potassium, and slightly greater than sodium and chlorine. A 70 kg (150 lb) human body contains about 140 grams of sulfur.
In plants and animals, the amino acids cysteine and methionine contain most of the sulfur, and the element is present in all polypeptides, proteins, and enzymes that contain these amino acids. In humans, methionine is an essential amino acid that must be ingested. However, save for the vitamins biotin and thiamine, cysteine and all sulfur-containing compounds in the human body can be synthesized from methionine. The enzyme sulfite oxidase is needed for the metabolism of methionine and cysteine in humans and animals.
Disulfide bonds (S-S bonds) between cysteine residues in peptide chains are very important in protein assembly and structure. These covalent bonds between peptide chains confer extra toughness and rigidity.[74] For example, the high strength of feathers and hair is due in part to the high content of S-S bonds with cysteine and sulfur. Eggs are high in sulfur to nourish feather formation in chicks, and the characteristic odor of rotting eggs is due to hydrogen sulfide. The high disulfide bond content of hair and feathers contributes to their indigestibility and to their characteristic disagreeable odor when burned.
Homocysteine and taurine are other sulfur-containing acids that are similar in structure, but not coded by DNA, and are not part of the primary structure of proteins. Many important cellular enzymes use prosthetic groups ending with -SH moieties to handle reactions involving acyl-containing biochemicals: two common examples from basic metabolism are coenzyme A and alpha-lipoic acid.[74] Two of the 13 classical vitamins, biotin, and thiamine, contain sulfur, with the latter being named for its sulfur content.
In intracellular chemistry, sulfur operates as a carrier of reducing hydrogen and its electrons for cellular repair of oxidation. Reduced glutathione, a sulfur-containing tripeptide, is a reducing agent through its sulfhydryl (-SH) moiety derived from cysteine. The thioredoxins, a class of small proteins essential to all known life, use neighboring pairs of reduced cysteines to work as general protein reducing agents, with similar effect.
Methanogenesis, the route to most of the world's methane, is a multistep biochemical transformation of carbon dioxide. This conversion requires several organosulfur cofactors. These include coenzyme M, CH3SCH2CH2SO3?, the immediate precursor to methane.[75]
Metalloproteins and inorganic cofactors
Inorganic sulfur forms a part of iron–sulfur clusters as well as many copper, nickel, and iron proteins. Most pervasive are the ferrodoxins, which serve as electron shuttles in cells. In bacteria, the important nitrogenase enzymes contains an Fe–Mo–S cluster and is a catalyst that performs the important function of nitrogen fixation, converting atmospheric nitrogen to ammonia that can be used by microorganisms and plants to make proteins, DNA, RNA, alkaloids, and the other organic nitrogen compounds necessary for life.[76]
https://en.wikipedia.org/wiki/Sulfur
jmowreader
(50,557 posts)"Organic" to a food person (grown according to the standards of organic farming, which pretty much means no synthetic chemicals on plants and no antibiotics on animals) is different from organic to a chemist.
To a chemist, any "organic" compound must contain carbon. There are a few inorganic carbon compounds (CO2, carbonates and cyanides are the big three) but in general if it's a compound that contains carbon, it's an organic compound.
There is no "periodic table of the compounds," only one of elements. Therefore, the whole periodic table is inorganic.
Quixote1818
(28,936 posts)Quixote1818
(28,936 posts)Snip: (Sulfur is listed)
All of the objects are considered organic except the rocks and the paddle boat. To a chemist, the term organic describes chemical compounds that contain carbon and other elements such as hydrogen, oxygen, nitrogen, sulfur, or phosphorus. For example, the pain reliever acetaminophen is organic and its chemical formula is C8H9NO2.
At the grocery store, the term organic describes foods raised under specific conditions. For example, beef labeled organic is from cows that were not given antibiotics, growth hormones, or fed animal by-products.
All of the organisms pictured are alive and composed of organic compounds.
Then why is sugar organic? Let’s find out more.
https://www.texasgateway.org/resource/organic-or-inorganic
mopinko
(70,103 posts)approved organic pesticides.
Quixote1818
(28,936 posts)The article is referring to the FDA's definition of organic pesticides as defined in agriculture, not organic vs inorganic chemistry.
RockRaven
(14,966 posts)towards the face for ease of eating? These are the important observations about bananas, after all.
Quixote1818
(28,936 posts)Hee Hee!
KT2000
(20,577 posts)the chemical industry has successfully fought off testing for and accounting for effects such as endocrine disrupting chemicals. In some cases higher doses are safer than lower doses. Testing is being done but not by the industry - at least they are not sharing their research.
The dose makes the poison is outdated as we learn more about the effects of chemical exposures. Also it is not honest to compare for example, an apple that is made up of many chemicals with a single chemical. Mixtures behave differently than exposure to a single chemical.
I have seen this sort of thing being used by an ag university prof to get our little hamlet to stop our no spray on roadside policy. It was laughable as most people realized. We are still using mowers instead of the more highly toxic herbicides our state uses elsewhere.